Psilocybin is more than a mind-altering drug
Psilocybin is best known for its profound influence on consciousness, emotions, and spirituality. However, psilocybin's effects don't just occur in the brain. Once ingested, psilocybin first enters the intestines, where it is converted into psilocin. This is the active ingredient that not only affects serotonergic receptors in the brain, but also interacts with the intestinal wall, the immune system and the microbiome.
We also wrote earlier that psilocybin also has anti-inflammatory properties in preclinical research and may even play a role in slowing down aging and protecting DNA (research on cells in vitro and mice). If these effects are also present in humans and are combined with a healthy lifestyle, a powerful synergy It's possible that mental, physical, and cellular processes reinforce each other. We use these scientific insights to further explore and reason about the impact of psilocybin on the microbiome. Use the buttons to read more about these mechanisms of action of psilocybin.
In this article, we'll explain the individual effects of psilocybin on the microbiome, inflammation, aging, and lifestyle. We'll also explain how these effects interact. We'll also discuss the potential advantages and disadvantages, so that a complete picture emerges of what psilocybin can do in the body, especially the intestines.
From psilocybin to psilocin: the first stop is the gut
Psilocybin is a natural compound found in certain mushrooms and truffles. It belongs to the group of tryptamines, a family of molecules that resemble the body's own tryptamine and serotonin.
When a person takes psilocybin, it is rapidly converted into psilocybin in the gastrointestinal tract and liver. psilocin. This conversion occurs by enzymes that cleave off a phosphate group – hence psilocybin is also called the “prodrug” of psilocin.
- Psilocybin = inactive form (as found in truffles).
- Psilocin = active form that affects the body and brain.
Psilocin can bind to serotonin due to its similarity serotonin receptors in the nervous system. In the brain, the main 5-HT2A receptor important, because its activation is related to the psychedelic effects (visual changes, more intense emotions, sense of unity).
But psilocin isn't confined to the brain: it's also found in the intestines Correctly, it affects both host cells and microorganisms. Also compare the structures of serotonin, psilocybin, and psilocin and imagine how these substances can fit onto the same receptors due to their similar shapes.
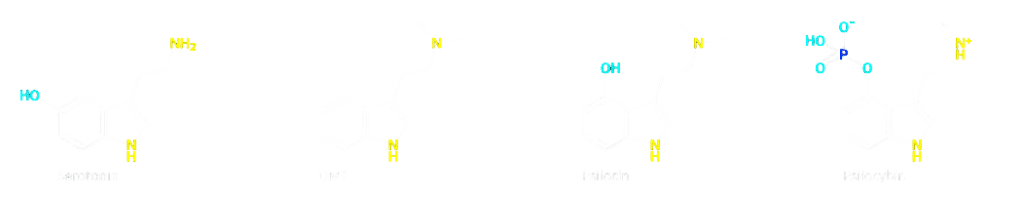
Serotonin in the gut: more than a neurotransmitter
What is serotonin?
Serotonin (5-hydroxytryptamine, 5-HT) is a neurotransmitter that plays a role in mood, sleep, and impulse control in the brain. But less well known is that approximately 90% of all serotonin in our body is produced in the intestines by special cells in the intestinal wall, the enterochromaffin cells.
In the intestine, serotonin has several functions:
- It regulates the peristalsis (the squeezing movements that move food through the intestines).
- It affects the sensitivity of the intestine (pain, pressure, stretch).
- It communicates with the immune system in the intestinal wall.
It works like signaling substance for microbes in the intestinal lumen.
Serotonin as communication between humans and microbes
Microorganisms in the intestine are not just passive inhabitants, but are in constant communication with our bodies. They can produce substances that affect humans (for example, fatty acids like butyrate), but they can also recognize and respond to human substances.
Serotonin is one such signaling substance. Some bacteria have specific proteins that allow them to detect serotonin and adjust their behavior accordingly. Psilocin, which closely resembles serotonin, likely exerts its effects via the same mechanisms.
Which microorganisms respond to serotonin (and possibly psilocin)?
There are two known ways in which microorganisms in the microbiome can measure and respond to the levels of serotonergic substances such as serotonin and the similar substance psilocin: the CpxA/CpxR system in bacteria and the LasR system.
The CpxA/CpxR system
It CpxA/CpxR system is a so-called two-component system which is common in Gram-negative bacteria such as Escherichia coli and Salmonella. CpxA is a membrane-bound sensor that detects changes in the environment. When serotonin, or possibly psilocin (further research needed), binds to CpxA, signal transmission to the regulator CpxR changes. This changes genes involved in virulence (the pathogenic potential of a bacterium) is suppressed. The result is that the bacterium is less able to adhere, produces fewer toxins, and behaves less aggressively in the intestine. In this way, an increased presence of serotonergic substances can protective effect have against excessive bacterial load.
Microorganisms involved:
- Escherichia coliThe most common intestinal resident: usually harmless, but some strains cause diarrhea, urinary tract infections, or even blood poisoning. Through CpxA/CpxR, they respond to serotonin/psilocin with less virulence.
- Salmonella enterica: Responsible for food poisoning and typhoid. Through CpxA, serotonin can inhibit the activity of so-called virulence islands, thus hindering the bacteria's ability to spread.
- Shigella spp: Causes bloody diarrhea (shigellosis). Although less well-researched, serotonin is also expected to inhibit virulence here.
- Yersinia spp: Known from the plague (Y. pestis), but also intestinal infections (Y. enterocolitica). The CpxA system is influenced by serotonin, likely with inhibitory effects on pathogenic behavior.
- Klebsiella pneumoniaeNormally present in the intestines, but in weakened individuals it can cause pneumonia or sepsis. CpxA/CpxR helps this bacterium process stress signals; serotonin reduces virulence.
- Enterobacter spp: Common intestinal commensals that are usually harmless. They can, however, cause infections in weakened immune systems. Here too, serotonin likely has a mitigating effect on virulence.
- Citrobacter spp: Similar to Enterobacter: present in the intestines, sometimes causing opportunistic infections. CpxA makes them sensitive to serotonin, making them less aggressive.
The LasR system
The LasR system is mainly used by Pseudomonas aeruginosa, an opportunistic bacterium. LasR is part of a quorum-sensing mechanism: a way in which bacteria communicate with each other to adjust their behavior when they are present in large numbers. Normally, LasR binds to auto-inducer molecules that bacteria produce themselves, but research shows that serotonin disrupts this system in P. aeruginosa can activate. This leads to an increase in virulence: the bacteria form more biofilm (a protective slime layer), produce more harmful enzymes, and become more aggressive. This has not yet been directly investigated for psilocin, which closely resembles serotonin, but it is plausible.
Microorganisms involved:
- Pseudomonas aeruginosa: An opportunistic pathogen found in the intestines, skin, and lungs, it is particularly dangerous in weakened individuals. Through the LasR system, this bacterium responds to serotonin by increasing its virulence. It forms more biofilm and releases more harmful enzymes, making infections more severe and difficult to combat. A similar effect is suspected for psilocin, but this remains to be confirmed.
- Pseudomonas fluorescensUsually a relatively harmless organism, but can become opportunistically pathogenic under certain circumstances. This species possesses LasR homologs and could theoretically react similarly, but this has not yet been convincingly demonstrated.
- Burkholderia cepacia complexKnown as a cause of persistent infections in people with cystic fibrosis. This group of bacteria has several quorum-sensing regulators related to LasR. It is conceivable that serotonin or psilocin plays a role in this, but firm research results are lacking.
- Acinetobacter baumanniiA major cause of hospital infections, particularly in the respiratory tract, urinary tract, and wounds. This bacterium also has LasR-like regulators, but whether serotonin or psilocin actually increases virulence in this case has not yet been convincingly proven.
Net effect on the microbiome
This piece about the positive effect of psilocybin on the microbiome is our hypothesis, based on extrapolation from studies of psilocybin on human cells and animals. For now, this remains a hypothesis because the direct effects of psilocybin on the composition of the human microbiome have not yet been convincingly demonstrated by scientific studies.
On the one hand, psilocin can be a health benefit mean. In bacteria that use the CpxA/CpxR system, such as Escherichia coli, Salmonella and Klebsiella pneumoniae, the binding of serotonin or psilocin leads to a decrease in virulence. This means that these bacteria adhere less effectively to the intestinal wall, produce fewer toxins, and behave more calmly. This can be positive for the host, as the burden on the immune system is reduced and the risk of inflammation is reduced. Yeasts such as Candida albicans react favourably: serotonin and probably also psilocin inhibit the transition to the aggressive hyphal form and thus limit the risk of overgrowth.
On the other hand, psilocin can also be a unfavorable effect have. Bacteria that use the LasR system, such as Pseudomonas aeruginosa, may actually respond with an increase in virulence. Under the influence of serotonin and psilocin, they form more biofilm and produce harmful enzymes. This makes them more difficult to combat and can lead to increased inflammation and symptoms, especially in people with weakened immune systems or a disrupted microbiome. Other bacteria with LasR-like systems, such as Burkholderia cepacia and Acinetobacter baumannii, show similar responses.
It is therefore clear that psilocin is not unequivocally "good" or "bad" for the intestinal flora. The effect depends on the present microorganisms and the balance of the microbiome. In healthy people with stable gut flora, the beneficial effects may predominate, while in people with many opportunistic bacteria, the unfavorable effects may be more noticeable.
However, this also needs to be put into perspective. A psilocybin experience lasts on average six o'clock. This means that the exposure of microbes to psilocin is relatively short. For most microorganisms, the impact will be temporary and will not permanently change the composition of the intestinal flora. At most, there may be some increased or decreased activity, which then decreases again as the drug leaves the body.
In summary, psilocybin could theoretically contribute to a reduction in the number of microbes less pathogenic behavior, which is positive for health, while for other microbes it is a stimulating effect may have a temporary detrimental effect. Given the short duration of a session, the impact on the microbiome is likely limited. Further research should clarify the exact influence of psilocin on the LasR system and CpxA/CpxR system. We hope and expect that the effect on the CpxA/CpxR system is greater than on the LasR system, as this would better explain the positive effects of psilocybin on lasting mood improvements.
Psilocybin, microbiome, inflammation, aging and lifestyle: one connected whole
When we compare the various effects of psilocybin, a clear pattern emerges: the microbiome is the linchpin that connects everything. Psilocybin's influence on gut microbes impacts inflammation, cell aging, and even the ability to make and maintain healthy lifestyle choices.
One of the most important effects in the intestines is that psilocybin is absorbed by many microorganisms through the CpxA/CpxR system the virulence decreases.This literally gives the immune system a rest and ensures a lower ignition pressure.
Less inflammation in the intestines translates into a healthier microbiome environment. A calmer gut flora produces more protective substances such as short-chain fatty acids, which in turn strengthen the intestinal wall and further inhibit inflammation. This cascade affects the rest of the body: lower inflammation levels mean less damage to cells and DNA. Chronic inflammation is, after all, a significant accelerator of aging, partly because it causes telomeres, the protective caps on our DNA, to shorten more quickly. By inhibiting inflammation and reducing cellular stress, psilocybin can indirectly contribute to healthy aging.
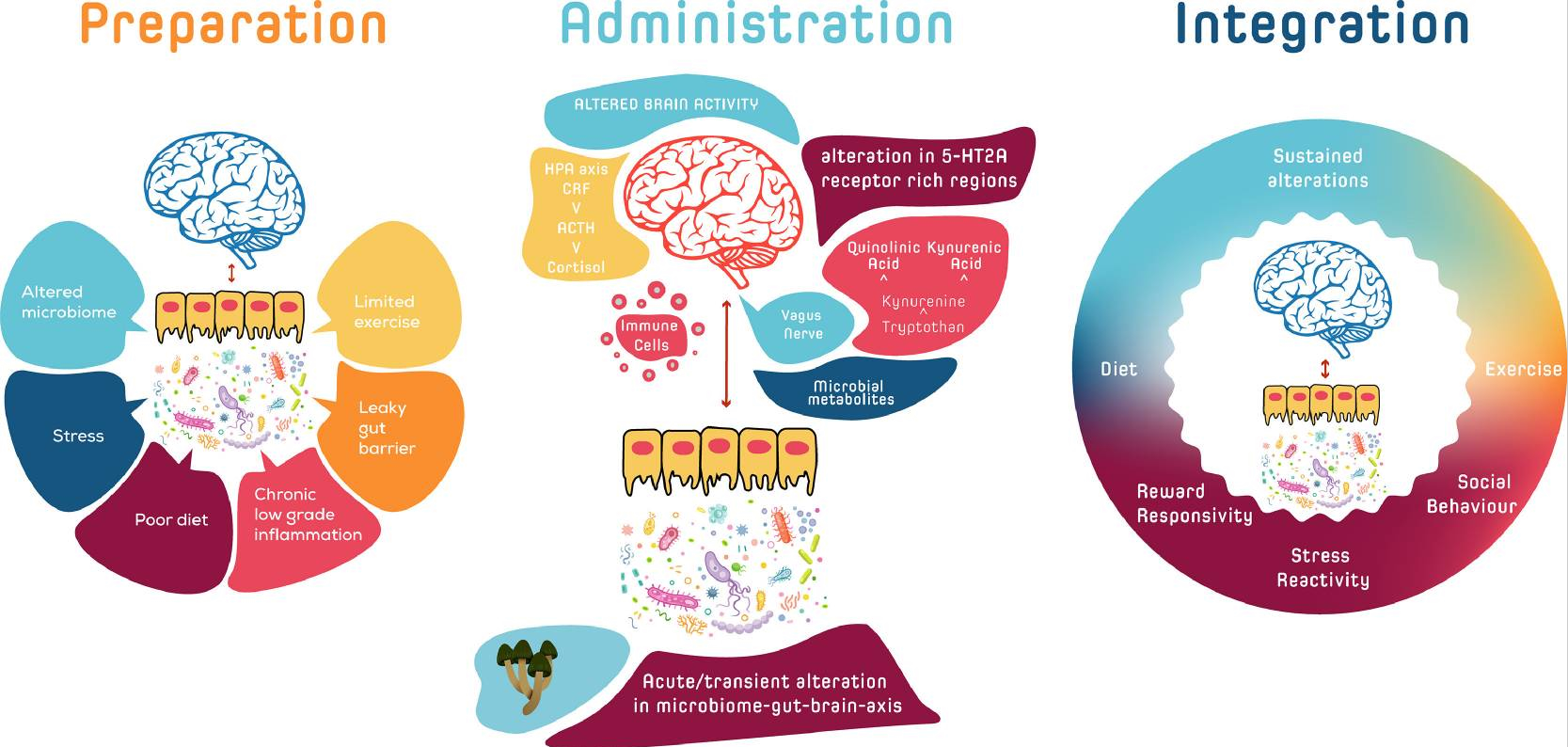
This creates a synergistic whole: the microbiome influences inflammation, inflammation influences aging, and psilocybin (psilocin) acts on all these levels simultaneously. The psychological effects of psilocybin also play a role. During a session, people often gain new insights and greater motivation to live healthier. This behavioral change has a direct impact on the microbiome: a higher-fiber diet, more exercise, better sleep, and less stress strengthen the gut flora, further supporting the anti-inflammatory and anti-aging effects of psilocybin.
Thus, psilocybin forms a bridge between mind and body. In the gut, it influences microbes and inflammation levels; in the brain, it creates space for new patterns and behavioral choices. Both pathways reinforce each other via the microbiota–gut–brain axis. Ultimately, a vicious cycle of positive change emerges: a calmer microbiome leads to less inflammation, which slows cell aging and paves the way for sustainable lifestyle improvements, which in turn further strengthen the microbiome.
Conclusion and sources
The potential positive effects of psilocybin on the microbiome don't occur in isolation. They form the starting point of a chain reaction that affects inflammation, aging, and behavior. It is this holistic connection that explains why psilocybin can not only bring mental relief but could also have tangible physical health benefits.
At the same time, it is important to emphasize that many of these findings so far have mainly come from preclinical research (cells and mice) come. Large-scale and long-term human research is still needed to confirm the extent to which psilocybin actually has anti-inflammatory, anti-aging or microbiome-modulating effects.
What is already clear is that psilocybin forms a powerful bridge between mind and body. It can provide insights and motivation to live healthier, and precisely these lifestyle changes have a proven positive effect on gut flora, inflammation, and aging. This creates a synergy between psychological, biological, and lifestyle processes.
This blog post is based on our own articles and insights, supplemented with scientific sources. The buttons below will guide you through the physical effects of psilocybin, the research we cite, and how to register for psilocybin therapy.
